 Dichlorphenamide increases potassium excretion and can cause hypokalemia and should be used cautiously with other drugs that may cause hypokalemia including laxatives. While lubiprostone may cause diarrhea as a side effect, drug discontinuation alone may resolve the diarrhea.ĭichlorphenamide: (Moderate) Use dichlorphenamide and lubiprostone together with caution. In general, it would be illogical to concurrently administer crofelemer in combination with lubiprostone. Monitor serum potassium levels to determine the need for potassium supplementation and/or alteration in drug therapy.Ĭalcium Phosphate, Supersaturated: (Moderate) Sodium phosphate monobasic monohydrate sodium phosphate dibasic anhydrous should not be combined with additional laxatives or purgatives when being used to evacuate the bowel prior to colonic radiologic examinations or surgery.Ĭrofelemer: (Moderate) Crofelemer, through its local effects as an antidiarrheal on chloride channels in the intestine, may theoretically antagonize the pharmacologic effects of lubiprostone. 
In general, it would be illogical to concurrently administer antidiarrheals in combination with lubiprostone.īumetanide: (Moderate) Loop diuretics may increase the risk of hypokalemia especially in patients receiving prolonged therapy with laxatives. The clinical significance of these potential interactions is uncertain.īelladonna Opium: (Moderate) Opium can decrease GI motility and may antagonize the effects of drugs used to treat constipation, such as lubiprostone. 
However, lubiprostone may cause diarrhea as a side effect, but drug discontinuation alone may resolve the diarrhea.Īnticholinergics: (Moderate) Antimuscarinic drugs can promote constipation and pharmacodynamically oppose the action of drugs used for the treatment of constipation, such as lubiprostone. In general, it would be illogical to concurrently administer antidiarrheals in combination with lubiprostone. Patients should be aware of the risk of syncope and hypotension during treatment.Īlosetron: (Moderate) Alosetron can decrease GI motility and may antagonize the effects of drugs used to treat constipation, such as lubiprostone. Several cases reported concomitant use of medications known to lower blood pressure, which may increase the risk for the development of syncope or hypotension. Although syncope and hypotension generally resolved following lubiprostone discontinuation or prior to next dose, recurrence has been reported with subsequent doses. Concomitant diarrhea or vomiting was noted in some patients prior to developing the adverse reaction. Most cases occurred in patients taking 24 mcg twice daily and some occurred within an hour after taking the first dose or subsequent doses of lubiprostone. Cases of syncope and hypotension, some requiring hospitalization, have been reported during postmarketing experience with lubiprostone. Patients who experience diarrhea and vomiting with lubiprostone have an increased risk of syncope and hypotension. 
Patients should be informed of this side effect and should notify their health care provider if the diarrhea becomes severe. 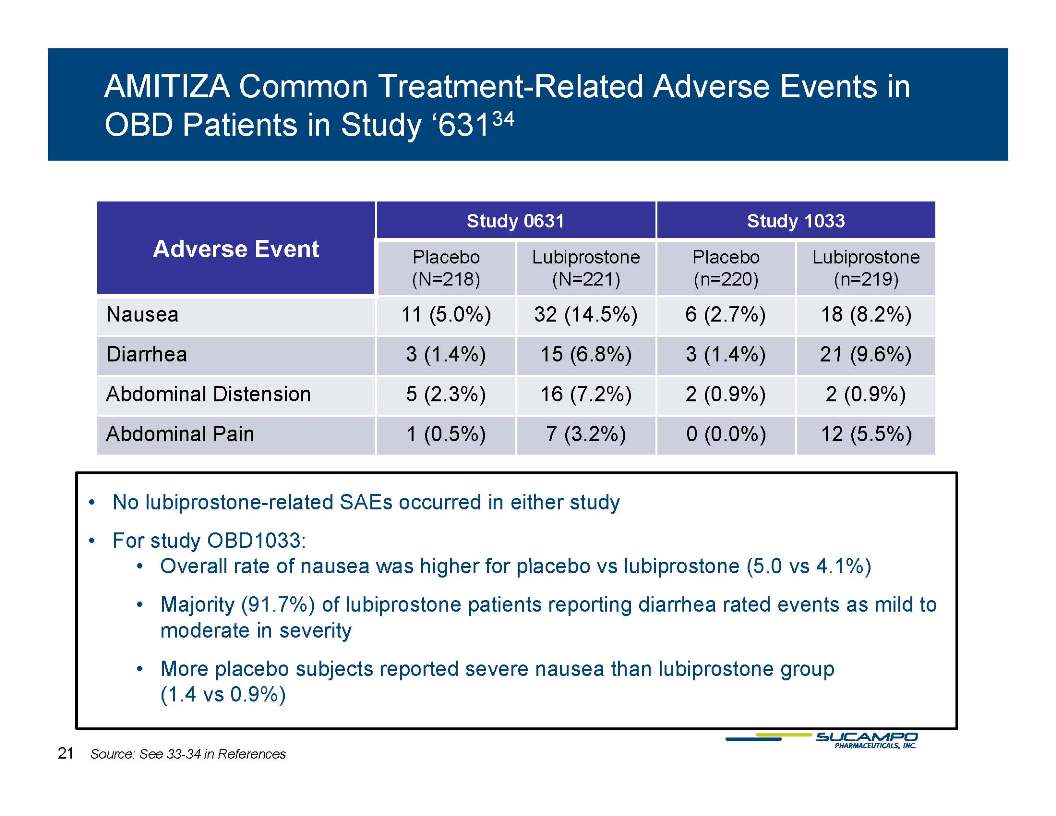
Diarrhea is a common adverse effect of lubiprostone. Lubiprostone should not be administered to patients with severe diarrhea. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
